- Drug Spotlight, Home Featured
- Home Featured, Latest News


- Home Featured, Latest News


- Home Featured, Latest News
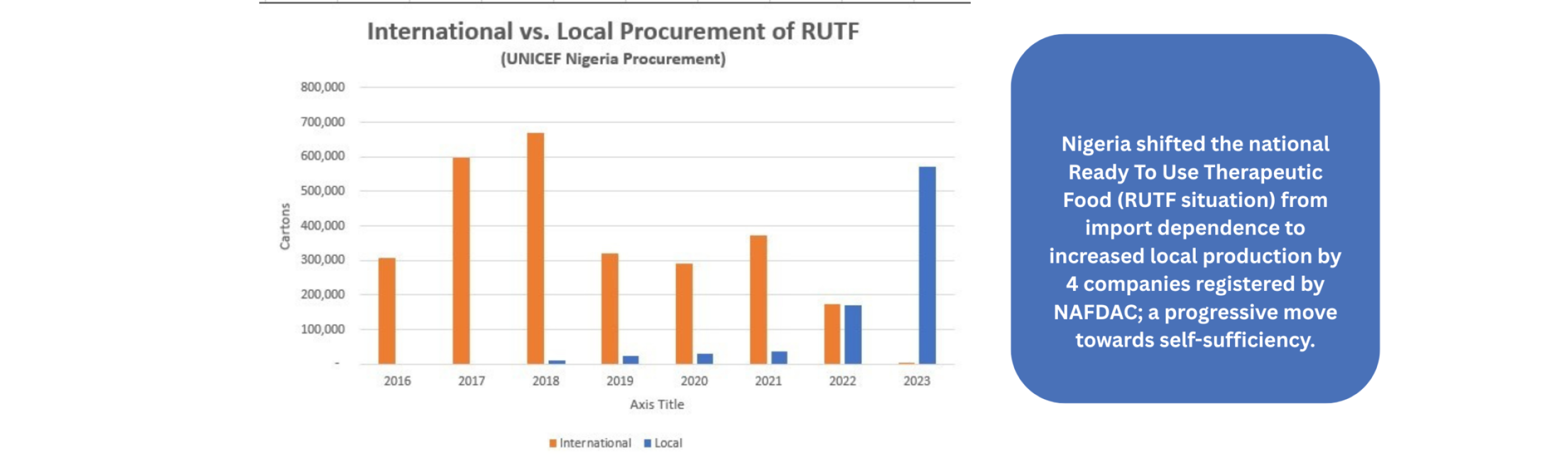
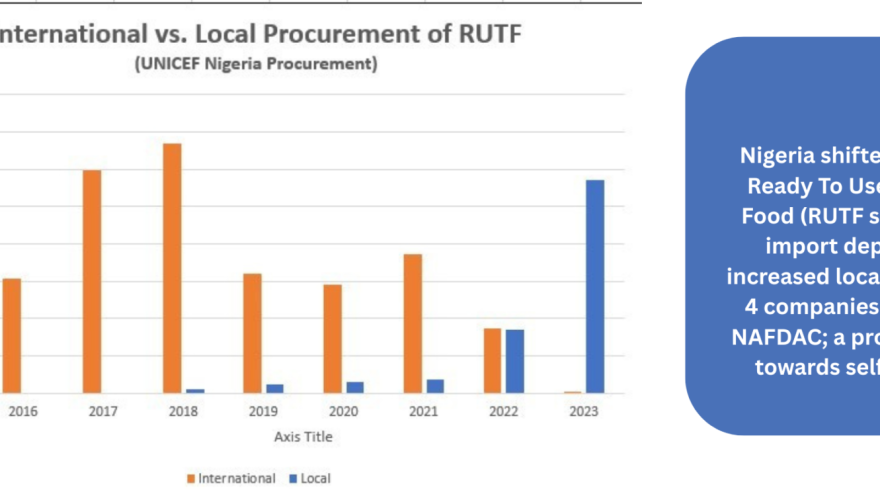
- Food Spotlight, Home Featured
Launch of the Implementation of Strategy and Roadmap for Trans-fatty…
Peer Reviewed Publications:
- Adeyeye, Mojisola, John Kayode, and Sabitu Olasupo. Traceability for Strengthening Supply Chain Systems and Enhancing Real-Time Visibility: Focus of NAFDAC on Advancing Vaccines Traceability in Nigeria. Ann. Acad. Med. Siles. 2024;78:276-281
- Mojisola Christianah Adeyeye, Uche Sonny-Afoekelu, Edosa Ogbeide, Abayomi Tosin Akinyemi, Ake Paul Ayodele, Olufemi Balogun & Sabitu Babatunde Olasupo (2026). Web-based Dossier Management System (DMS) and Emerging Technologies to Facilitate Faster Registration and Access to Human Drugs: NAFDAC’s Experience: Ther Innov Regul Sci (2026).
- Aderibigbe, O. O., Adamu, B. B., Osaji, I. O., Babarinde, A. O., Nwachukwu, C. U., Adegboye, A., … & Adeyeye, M. C. (2023). Secondary Validation of a Liquid Scintillation Counter-method for Residues of Tetracyclines, Beta-lactams, and Sulfonamides in Seafood/Aquaculture Products. Journal of Food Protection, 86(4), 100055.
- Adeyeye, M., Olasupo S., Akinyemi, A., Kayode, J., & Sonny-Afoekelu, U (2024). Impact of Digitalization in strengthening the regulatory functions of NAFDAC: Opportunities and Challenges for other National Regulatory Authorities. Journal of Regulatory Science, 12(1). Partly funded by Internally Generated Revenue, Global Fund, BMGF, WHO& USAID
- Kanu, N. M., Adeyeye, C. M., Abiola, V. O., Adekunle–Segun, O., Adegoke, E. M., Akapo, A., & Okezue, M. A. (2024). The safety, quality evaluation, and lot release of COVID-19 vaccines imported and used in Nigeria from march 2021 to march 2022. Discover Health Systems, 3(1), 15. Funded by Internally Generated Revenue
- Adeyeye, M., Kayode, J., Adeniran, A., Osho, F., & Udokwelu, W. (2023). Enabling Pharmaceutical Traceability in The Nigerian Supply Chain using GS1 Global Standards: Lean Traceability Including In-Country Serialization of COVID-19 Vaccines. Journal of Regulatory Science, 11(1), 1-14. Partly funded by Internally Generated Revenue, Global Fund, BMGF, WHO& USAID
- Elemuwa, U. G., Bitrus, F., Oreagba, I. A., Osakwe, A. I., Abiodun, A. S., Onu, K., … & Adeyeye, C. M. (2024). Trends in Adverse Event Reporting Before and After the Introduction of the Med Safety App in Nigeria. Pharmaceutical Medicine, 1-9.
Recalls and Safety Alerts
These are recalls and safety alerts released by NAFDAC. The alert notices and safety communications contain safety measures to be taken and information that may impact both treatment and diagnostic choices for healthcare providers and patients.
Watchlist & Blacklist of Companies
The Watchlist & Blacklist of Companies page provides an official and regularly updated record of companies that have been identified by NAFDAC as non‑compliant with regulatory standards. It highlights organizations that have engaged in serious or repeated violations, including poor manufacturing practices, submission of falsified documents, quality defects, and breaches of importation or licensing requirements.
Market Authorization Withdrawal
The National Agency for Food and Drug Administration and Control (NAFDAC) hereby inform the general public that the products listed on this page have been officially withdrawn, suspended, or cancelled by the Agency either for commercial reasons or due to deregistration.
NAFDAC Updates
Our Services
Meet the Director General.
Prof Moji Adeyeye is the Director General of National Agency for Food and Drug Administration and Control (NAFDAC) where she has added strong governance structure, regulatory system strengthening, and strengthening of the local pharmaceutical companies through international best practices with emphasis on local manufacturing.

Prof. Moji Christianah Adeyeye
Director General, NAFDAC