Products We Regulate
Our Services
- Product Registration and Registration
- Clinical Trail
- NAFDAC Greenbook (Registered Drug Database)
- Registered Products (non drug) Database
- NAFDAC eLicense
- List Item #1
- List Item #2
- List Item #3
- List Item #1
- List Item #2
- List Item #3
Meet the Director General
Prof Moji Adeyeye is the Director General of National Agency for Food and Drug Administration and Control (NAFDAC) where she has added strong governance structure, regulatory system strengthening, and strengthening of the local pharmaceutical companies through international best practices with emphasis on local manufacturing.

Prof. Moji Christianah Adeyeye
Director General, NAFDAC
Our Services
- Product Registration and Registration
- Clinical Trail
- NAFDAC Greenbook (Registered Drug Database)
- Registered Products (non drug) Database
- NAFDAC eLicense
- List Item #1
- List Item #2
- List Item #3
- List Item #1
- List Item #2
- List Item #3
Our Services
- Product Registration and Registration
- Clinical Trail
- NAFDAC Greenbook (Registered Drug Database)
- Registered Products (non drug) Database
- NAFDAC eLicense
- List Item #1
- List Item #2
- List Item #3
- List Item #1
- List Item #2
- List Item #3
NAFDAC becomes the 24th member of the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use
The National Agency for Food and Drug Administration and Control (NAFDAC), Nigeria, has officially been admitted as a Member of the International Council for Harmonisation of Techn
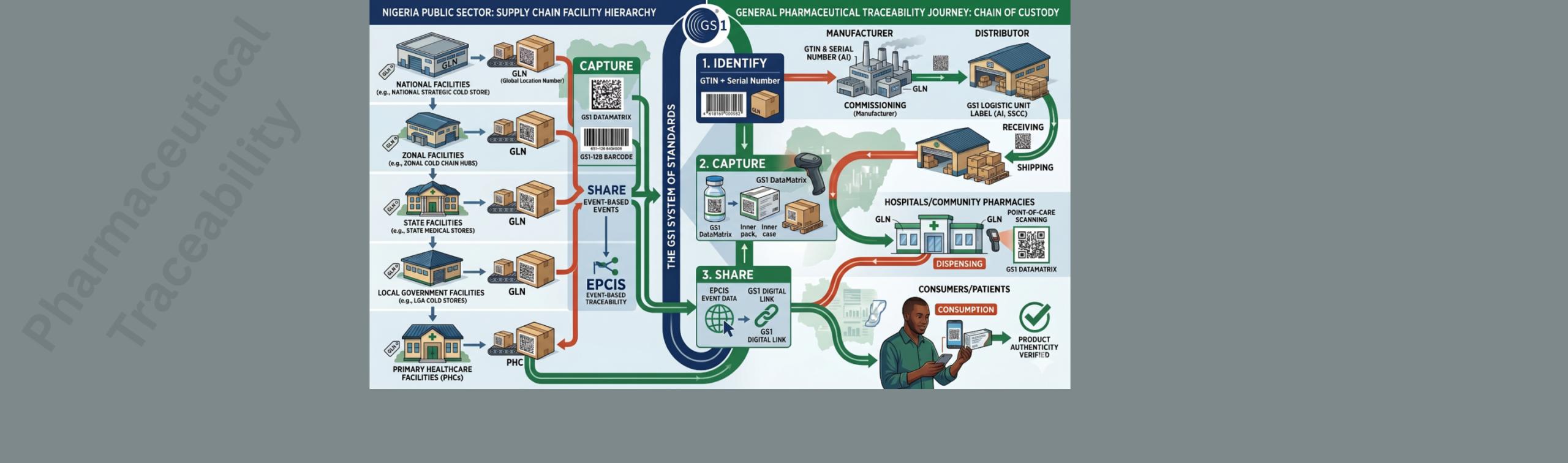
Pharmaceutical Traceability & Supply Chain Monitoring
Securing the Journey of Medicines — From Manufacturer to Patient Medicines that are diverted, expired, or simply unavailable when needed cost lives. Traceability — the ability
NAFDAC’s WHO ML3 Re-Benchmarking Success: Sustaining Regulatory Excellence
In the dynamic world of global health, the ability to ensure the safety, quality, and efficacy of medical products is the cornerstone of public trust. For the National Agency for F
Add Your Heading Text Here
Add Your Heading Text Here
Add Your Heading Text Here

About NAFDAC
- Product Registration and Registration
- Clinical Trail
- NAFDAC Greenbook (Registered Drug Database)
- Registered Products (non drug) Database
- NAFDAC eLicense
- Product Registration and Registration
- Clinical Trail
- NAFDAC Greenbook (Registered Drug Database)
- Registered Products (non drug) Database
- NAFDAC eLicense
- Product Registration and Registration
- Clinical Trail
- NAFDAC Greenbook (Registered Drug Database)
- Registered Products (non drug) Database
- NAFDAC eLicense
- Product Registration and Registration
- Clinical Trail
- NAFDAC Greenbook (Registered Drug Database)
- Registered Products (non drug) Database
- NAFDAC eLicense