The National Agency for Food and Drug Administration and Control (NAFDAC) has issued a notification to Embassy Pharmaceutical and Chemicals Ltd, Lagos (Holder of Registration Certificate) and Laborate Pharmaceutical India, the manufacturer of Embacef 125 Powder for Oral Suspension, to recall its product following a received complaint reporting the caking of two bottles of the reconstituted suspension after the first day. This complaint was reported at the NAFDAC state office in Ekiti State, Nigeria.
The investigation of the complaint confirmed the product’s poor quality and safety concerns, as stated by the complainant. As a precautionary measure, NAFDAC has requested the recall of the affected batch and other batches with similar issues from the market.
Embacef 125 Powder for Oral Suspension contains Cefuroxime Axetil, an antibiotic used to treat infections caused by sensitive bacteria. It is effective against conditions such as bronchitis, gonorrhea, and Lyme disease. It can also be used to treat infections of the skin, ears, sinuses, throat, tonsils, and urinary tract.
Risk Statement: The use of substandard antibiotics can lead to worsening health conditions and even death. It can also contribute to the development of antibiotic-resistant bacteria.
Product details
The details of the products are as follows.
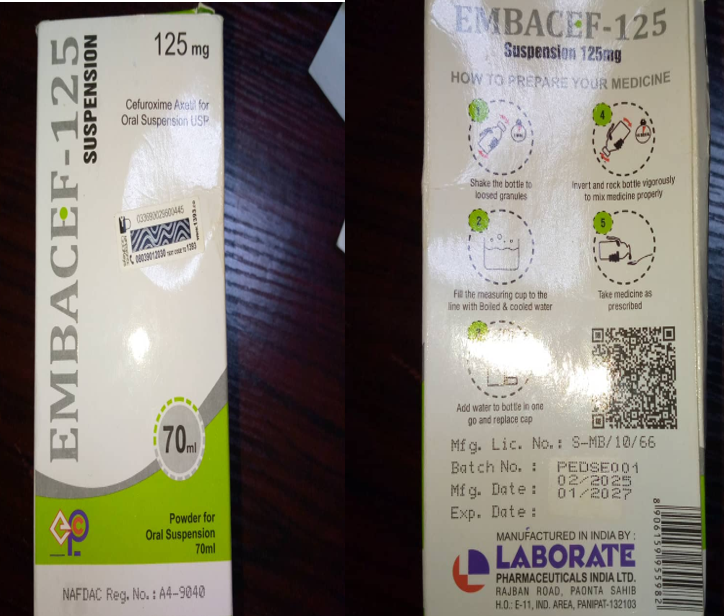
Product Name: Embacef- 125 Suspension (Cefuroxime Axetil USP Powder for Oral Suspension)
Holder of Registration Certificate: Embassy Pharmaceutical and Chemicals Ltd, Lagos
Product Manufacturer: Laborate Pharmaceutical India, E-11 Industrial Area, Panipat 132103.
Manufacturing date: 02/2025
Expiry date: 01/2027
Batch Number: PEDSE001
NRN: A4-9040
Product Image


All NAFDAC zonal directors and state coordinators have been directed to carry out surveillance and mop up the substandard product within the zones and states.
NAFDAC urges importers, distributors, retailers, healthcare providers, and patients to exercise caution and vigilance throughout the supply chain. This is essential to prevent the importation, distribution, sale, administration, or use of the recalled batch of the product. All medicinal products must be obtained from authorized/licensed suppliers. The products’ authenticity and physical condition should be carefully checked.
Healthcare professionals and consumers are advised to report any suspicion of substandard and falsified medicines to the nearest NAFDAC office, NAFDAC on 0800-162-3322 or via email: sf.alert@nafdac.gov.ng
Similarly, healthcare professionals and patients are also encouraged to report adverse events or side effects related to the use of the affected product to the nearest NAFDAC office, or through the use of the E-reporting platforms available on the NAFDAC website www.nafdac.gov.ng, or via the Med-safety application available for download on Android and IOS stores or via e-mail on pharmacovigilance@nafdac.gov.ng
Furthermore, note that this notice will be uploaded to the WHO Global Surveillance and Monitoring System (GSMS).
NAFDAC……….Customer-focused, Agency-minded!!!
